Floor plans, schools and neighborhood details might be what most parents have in mind when hunting for a new house, but Erin Rusling had a different focus: How long would an ambulance ride to the hospital take?
And so the mom of two settled on a house in Aledo—a 15-minute ride to Cook Children’s Hospital in Fort Worth—when moving her family from Oklahoma to Texas in 2022. That proximity gave her some peace of mind for when a seizure would inevitably strike her then 3-year-old daughter, Jane. Plus, Jane was already a patient at Cook Children’s, one of the only hospitals with highly specialized care for her rare and severe epileptic disease.

When it comes to rare diseases, defined as any condition affecting fewer than 200,000 people in the United States, an expert is not always in your backyard—never mind treatments or therapies, if they even exist. So for the Ruslings, a move to Texas was just another step on a journey that had already taken them all over the country.
They had driven five hours from Tulsa to Kansas City to meet the doctor who would finally diagnose Jane after an agonizing year-and-a-half of unanswered questions. They had Zoom calls with neurologists in California. They flew to Philadelphia for genetic research. And then, before their move, they drove four-and-a-half hours each way to Fort Worth every six months so that Jane could take part in a clinical trial.
Traveling, moving, searching—for answers, doctors, treatments—are all familiar to parents raising kids with rare diseases. And contradictorily, these rare parents, as they’re called, aren’t so rare at all. Although any one of these conditions may not affect many people, collectively, rare diseases affect 300 million people worldwide, and roughly one in 10 Americans, according to the National Organization for Rare Disorders (NORD). About half of them are children. For millions of moms and dads this means stepping outside of the life they envisioned and into roles as caregivers, case managers, advocates and fundraisers.
The Diagnostic Odyssey
Jane was just 4 months old when she had her first seizure. It was terrifying and confusing—it didn’t look like the full-body convulsions Rusling had always thought seizures were. Instead, baby Jane stared off to the side, shaking one arm up and down. Many tests later, doctors found nothing out of the ordinary. “They said it’s probably nothing, and it probably won’t happen again,” Rusling recalls.
But a few weeks later, it did happen again. And every few weeks, little Jane would have another seizure. At 16 months, she started having even more, up to two a week, and then small, atypical absence seizures began, sometimes 50 a day. Doctor after doctor was puzzled. “They were like, ‘We’re not sure what to give her. Maybe this? Maybe that?’” Rusling remembers. “And I was like, ‘That’s not good enough. We need to know what’s going on.’”
Finally, when she was almost 2, Jane was diagnosed with Dravet syndrome, a genetic epilepsy that gives rise to seizures that don’t respond well to medication.
The excruciating wait for answers goes on much longer for many families. On average, it takes at least five years for a person with a rare disease to get the correct diagnosis, according to NORD. It’s a journey that can be so prolonged—filled with research, testing, specialists and retesting—it’s often called the diagnostic odyssey.
“The time from onset of symptoms to diagnosis is significantly influenced by the clinician’s proficiency, informed by both their accumulated expertise and prior clinical encounters, as well as their comprehensive knowledge of pertinent medical literature,” explains Dr. Saima Kayani, a pediatric neurologist at Children’s Health in Dallas and associate professor at UT Southwestern.
While an estimated 80% of rare diseases are genetic in origin, there are nuances to finding the right diagnostic tools. “Assessments need to be tailored to detect potential systemic involvement,” she explains.
Kirstie Austin, who lives north of Fort Worth, remembers the frustration of knowing something was not right with her daughter Emma, but not finding any answers. At age 2, Emma began regressing—her speech was delayed and she was losing motor skills. Then came the outbursts, or as Austin says, “all-out raging”—episodes of screaming, hitting, biting and thrashing that would sometimes go on for an hour or more.
They took Emma to the pediatrician, a speech therapist, a chiropractor, even a dentist to correct a lip and tongue tie. There was bloodwork, testing for heavy metals, gut tests. “Every doctor was like, ‘We don’t see anything wrong,’” Austin recalls.
Then, sitting in her car seat one day while Austin drove down the highway, then 3-year-old Emma had a seizure. She was brought to the hospital and finally—with Austin’s insistence that something was really wrong—was offered genetic testing. At last, the Austins would get an answer, but it was one they didn’t want: Emma had Batten disease, a rare and fatal disease of the nervous system.
Unknown Territory for Parents
Processing that long-awaited diagnosis can take time. For rare parents, it’s the realization that their child may have a lifelong debilitating or life-limiting condition. They worry about access to treatments, supports and therapies—not to mention how they’ll pay for them. And they mourn the life they envisioned for their child.
“You start living in grief,” Austin says. “You’re looking at your child and it’s not just grieving for that child, it’s grieving things that you’re no longer going to see—her getting married or graduating high school, getting her driver’s license. All these little things you have in a normal life are stolen.”
A 2016 study that surveyed hundreds of rare parents found that nearly half of them were being treated for depression and anxiety. Frustration, fear and uncertainty were among the most frequent emotions these parents expressed.
RELATED: Special Needs Parents: What to Do When You’re Depressed?
The study also highlighted how incredibly isolating a rare disease diagnosis can be. More than half of the parents said their number of friends had reduced. What’s more, 75% of them had no contact with other parents with a child with a similar disease, and 42% had no access to a disease-specific support group.
Navigating the complex healthcare system can be daunting for these parents. And often, they feel they’re doing it on their own. “There’s no advocate who’s like, ‘What about this? Or what about that?’ It is up to the parents to talk to other parents, to look at clinical trials, to go to workshops online and to start putting the pieces together,” says Rusling.
And so rare parents often pour themselves into their child’s disease, becoming the experts themselves. They read medical journals and research papers, learn the ins and outs of genetics. They keep detailed records and push for new treatments.
“Your brain doesn’t shut down,” Rusling says. “When you go to bed at night, you’re sitting on your phone looking up clinical trials.”
Finding and Creating Support Systems
Amber Scuderi remembers deep diving into Trisomy 18, or Edwards’ syndrome, after her daughter Evalyn’s diagnosis. What she found, she says, was “a lot of doom and gloom.” Most experts agreed Evalyn wouldn’t live more than a few months at most. And so after a month-long stay in the NICU, Evalyn went home on hospice.

But Scuderi wasn’t convinced that research painted the whole picture. She found many statistics around Edwards’ syndrome were outdated. So even though she was told her daughter had only a 5–10% chance to make it to her first birthday, Scuderi pursued heart surgery, finding a doctor willing to repair her tiny heart at four months old.
Then Evalyn’s first birthday came and went, and Scuderi’s perspective shifted. “We had lived every day of our life waiting for her to pass away, expecting her to pass away. We spent a year grieving and then there was this mental shift of like, what do we do now?” Scuderi says. “How do we help her be functional in our family and in society? What can we do for her?”
Desperate to find out what Evalyn’s life might look like—and with nowhere to find the answer—Scuderi decided to fill the void herself. After joining the board of directors for Extra to Love, a DFW-based nonprofit supporting children with Edwards’ and Patau syndromes, Scuderi created a podcast for other parents. The point was to connect families and tell their stories—ones that weren’t always reflected in medical journals and doctor’s offices. She wanted to share that there were options—and hope.
RELATED: How to Help Friends Who Have Children with Special Needs?
From Mom to Advocate
Scuderi believes that sharing stories like Evalyn’s can help change attitudes about treatments that could help children with some rare diseases live longer lives. Heart surgery, for example, was shown in 2017 study to more than double the life spans of some babies born with Trisomy 13 or 18. But it’s still controversial for these children and not always given to parents as an option.
“There is a bad stigma that surrounds the diagnosis [of Trisomy 18] that they’re ‘incompatible with life.’ And therefore, why are you going to intervene on a child that’s incompatible with life?” she says. “And so we’re trying to remove the stigma.”
Rare parents know well what it’s like to fight for their kids. As Scuderi calls it, they have to “put on their advocacy hats.” It could mean searching for a medical team or hospital that supports their desire to pursue different treatments. Or it could mean convincing researchers and drugmakers that the disease is a good investment. Often, it’s a race against time.
That’s where the Austins find themselves as they navigate Emma’s disease. Currently, there is just one FDA-approved treatment for CLN2 Batten disease, an enzyme replacement therapy Emma receives every two weeks through a surgically placed port in her brain.
It’s not a cure, but rather an effort to stall the progression of Batten; to extend Emma’s life beyond the current expectancy of 6 to 12. They’re grateful for this treatment—of the approximately 7,000 known rare diseases, only about 500 have an approved treatment, according to NORD—but they’re frustrated there’s not more.
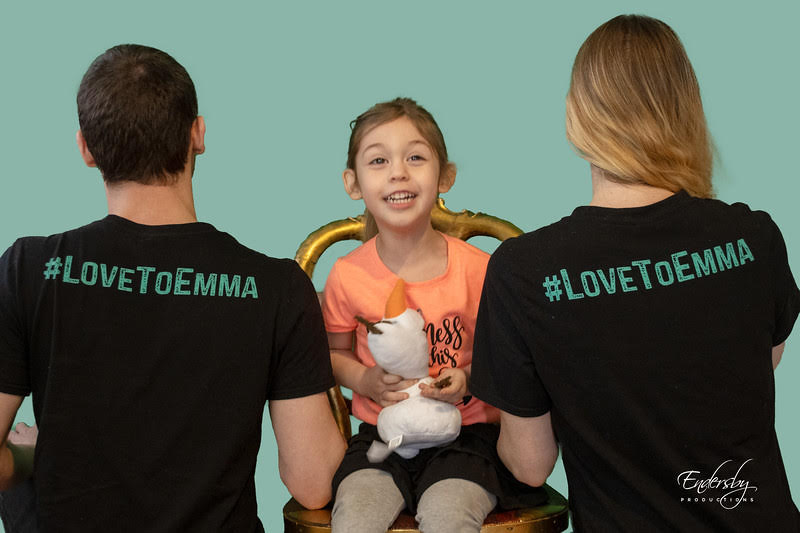
Then late last year, the Austins saw a glimmer of hope when researchers launched a clinical trial investigating gene therapy, treating just a single child with CLN2 Batten disease. “It came out of nowhere and it was this huge string of hope basically that they were dangling in our face,” says Austin. But just as quickly, the trial was halted, the biotech company citing “the challenging global economic market.”
It felt like a gut-punch to Emma’s parents. “We feel like our children are not profitable enough. It’s maddening,” says Austin. “We’re running out of time.”
Now advocating for accessible gene therapies, Austin says there are two in the pipeline for CLN2, both from companies seeking funding and partnerships to continue their research and development. With her GoFundMe, “Love to Emma – Cure Batten Disease,” Austin is raising awareness and funds to help her own family and the Batten community as a whole. But knowing there’s more power in numbers, she’s considering starting a nonprofit to pool families together to raise the kind of big funding needed for research.
“I’m trying to make my circle bigger and talk to strangers all over the globe. To advocate for something that I never knew anything about,” she says. “It’s a whole world that not enough people know about.”
And as she advocates for Batten disease research, Austin knows a breakthrough may not come in time to help Emma. “If that’s not our miracle we can’t give up,” she says. “We have to continue fighting.”
Top image courtesy of Amber Scuderi





